Ocean acidification— the hidden impact of global warming on our oceans
A pteropod shell is shown dissolving over time in seawater with a lower pH. When carbon dioxide is absorbed by the ocean from the atmosphere, the chemistry of the seawater is changed.
©National Oceanic and Atmospheric Administration
What is ocean acidification?
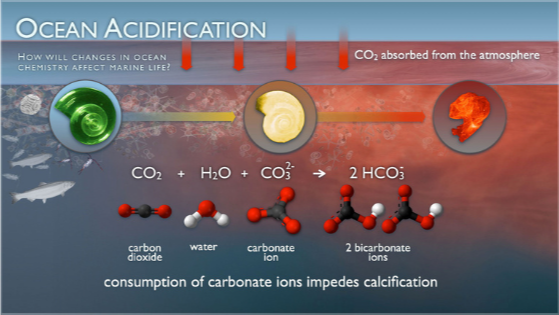
The ocean absorbs about 25–30% of the carbon dioxide (CO₂) humans release into the atmosphere. When CO₂ dissolves in seawater, it forms carbonic acid, which lowers the ocean’s pH. Since the Industrial Revolution, ocean acidity has increased by roughly 30% [1]. Acidity is measured using the pH scale, which ranges from 0 to 14. A pH of 0 is very acidic, 7 is neutral, and 14 is very basic or alkaline (the opposite of acidic). The ocean is naturally slightly basic, with a pH of about 8.2 [2].
Why does it matter?
Many marine organisms—like corals, shellfish, and plankton—depend on calcium carbonate to build shells and skeletons. Acidic water makes this process more difficult, leading to weaker shells, slower coral growth, and disruptions in food webs. Let’s imagine a tiny pteropod known as “sea butterfly” which would dissolve after 45 days in water with a pH projected for the year 2100 [1]. Further, ocean acidification affects fisheries, biodiversity, and coastal economies.
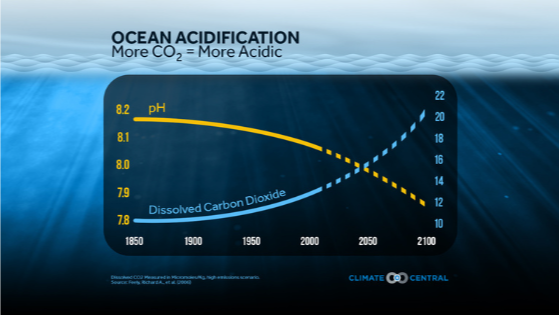
When CO₂ increases, the ocean becomes more acidic.
©2015 by Climate Central is licensed under CC BY 4.0
Connection to global warming
Ocean acidification and global warming share the same cause: excess CO₂ emissions. While CO₂ traps heat in the atmosphere (causing global warming), it also dissolves into oceans and changes water chemistry. Warmer oceans and acidification together create compounded stress for marine life. Warmer water can absorb less carbon dioxide than colder water, which slightly limits how much CO₂ dissolves in the ocean. However, this effect is not enough to offset the large amount of CO₂ released into the atmosphere by human activities, so ocean acidification continues.
The Baltic Sea – The Special Case
The Baltic Sea is especially vulnerable to ocean acidification. Unlike the open ocean, it has lower buffering capacity (alkalinity), meaning it cannot neutralise added acids as effectively. This makes even small increases in carbon dioxide have a stronger impact on pH. In addition, the Baltic Sea is shallow and semi-enclosed, with limited water exchange, which allows changes to build up more quickly.
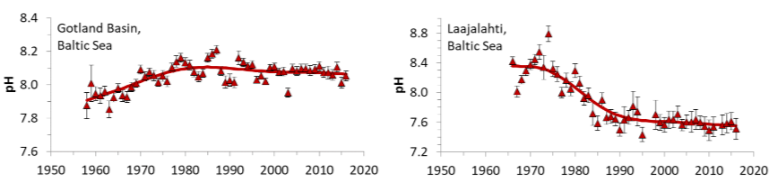
In practice, pH changes vary across the Baltic Sea because its buffering capacity is uneven, depending on river inputs, geology, and mixing with North Sea water. Northern areas with low alkalinity are more sensitive to acidification, while southern and more saline regions are better buffered. In addition, seasonal biology and pollution cause strong local fluctuations, making pH highly variable in both space and time [3].
Trends in pH estimated from monitoring data from the Gotland Basin and Laajalahti Bay near Helsinki.
©Carstenses and Duarte, 2019
Natural processes in the Baltic Sea also cause strong daily and seasonal pH fluctuations. During the day, photosynthesis by algae removes CO₂ from the water and increases pH, while at night and in deeper waters, respiration releases CO₂ and lowers pH again. These fluctuations can be quite large compared to the open ocean [4,5].
Human activities further increase the pressure. Nutrient pollution leads to algal blooms, and when this organic matter decomposes, it releases additional CO₂, contributing to acidification. As a result, the Baltic Sea experiences not only global climate-driven changes, but also local effects that intensify and complicate acidification.
What can we do in the Baltic Sea?
Reducing ocean acidification in the Baltic Sea starts with lowering CO₂ emissions, since this is the main driver of the problem. However, because the Baltic Sea is strongly affected by local conditions, regional actions are just as important. One key step is reducing nutrient pollution from agriculture and wastewater, which causes algal blooms. When these blooms decompose, they release CO₂ and further lower the pH. Improving water quality can therefore help limit acidification.
Protecting and restoring marine ecosystems—such as seagrass meadows and algae—can also help, because these organisms absorb CO₂ during photosynthesis and can locally increase pH. In addition, reducing overfishing and protecting biodiversity makes the ecosystem more resilient to environmental stress.
Although some natural processes in the Baltic Sea can partially buffer acidification, they are not sufficient to counteract rising CO₂ levels. This means that both global climate action and local environmental management are necessary to protect this unique and sensitive sea.